How caffeine works in the body
Cold Brew research published in this months issue of Roast Magazine

We're honored to of had some of our research on cold brew chemistry published in this month's issue of Roast Magazine.
The research which was independently conducted by our coffee scientist, Joseph Rivera, utilized high performance liquid chromatography (HPLC) to analyze differences between hot vs cold brewed coffee. Check out the article, it will sure surprise you.
The Chemistry of Brewing in your Cup
Organic Acids Revisited - Roast Magazine
June 1, 2015 - Check out this month issue of Roast Magazine where we revisit the topic of organic acids and provide a brief historical context of how this milestone research changed the specialty coffee industry. Download a PDF copy from our library downloads section.
The Chemistry of Organic Acids in Coffee: Part 3
Welcome back to this third and final issue of organic acids. In the last issue we briefly discussed the role of quinic, caffeic, and citric acid and its role in coffee’s flavor. This time we will explore acetic and malic acid and see how these seemingly simple acids play a major role in coffee’s complex flavor profile.
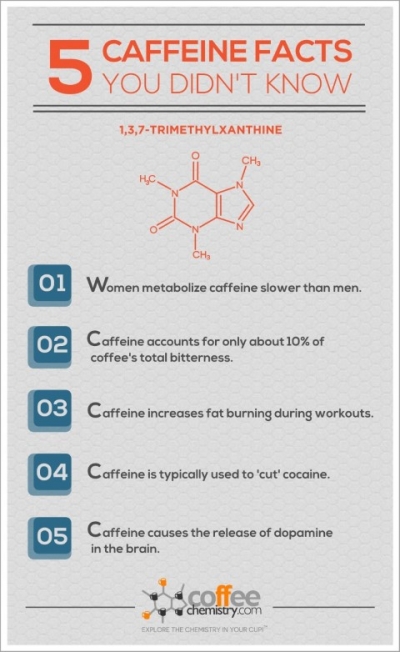
5 Caffeine Facts You Didn't Know
Think you know caffeine? Check out these five caffeine facts that will blow your mind.
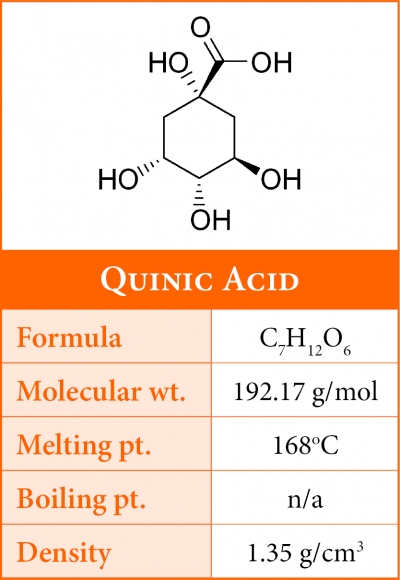
Quinic Acid
Quinic acid along with citric and malic represent a significant portion of coffee's total acid content. During roasting quinic acid progressively increases as the levels of chlorogenic acid decrease, suggesting that its formation results from the cleavage of the chlorogenic acid moiety.
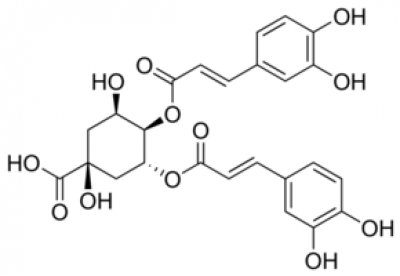
Chlorogenic Acid
Discovered in 1932, chlorogenic acids (CGA) represent a large family of esterified compounds present in green and roasted coffee. During roasting, CGA's slowly decompose to form caffeic and quinic acid with about 50% of the original CGA being destroyed in a medium roast.
Differences between Arabica and Robusta Coffee
Although arabica and robusta coffee may appear similar appearance - there are a number of differences that significantly differentiate these two popular species of coffee. The following list points out a few basic differences.