Displaying items by tag: green coffee
Cupping Fundamentals
Cupping is perhaps one of the most important skill anyone can develop within the coffee industry. Initially developed by green coffee buyer, "cupping" provides a consistent methodology for evaluating a coffee's quality before determining a price on a lot. In much the same way that a wine connoisseur would evaluate a fine Cabernet, cuppers must use a strict set of tasting protocols to assess quality.
Anatomy of a Coffee Bean
Perhaps the single most important factor in determining coffee quality is the care taken during post-harvest processing. A single mistake can have serious implications, at times, capable of spoiling en entire batch of coffee. But before we discuss the various processing methods, lets take a look at the coffee bean itself.
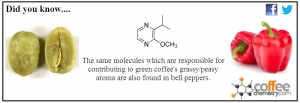
The Aroma of Green Coffee and Bell Peppers
That's right. These nitrogen containing pyrazines or specifically 3-isobutyl-2-methoxypyrazine are present in a wide range of food products including green coffee and bell peppers.
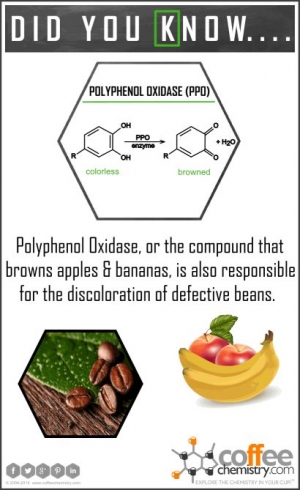
Polyphenol Oxidase
Washed Processed Coffee
Once coffee has been cultivated and transported to processing stations, the next step is to remove the outer pulp from the cherry, so that the bean itself can be extracted out. By far the vast majority of the coffee processed is via the washed process, where mechanical and biochemical processes are used to remove the outer layers surrounding the coffee bean.
Natural Processed Coffee
Typically in those countries such as Brazil where water is not readily accessible to farms, the dry process is the preferred method of processing. In this process, the coffee goes through a cursory separation to remove any debris then the cherries are set out on drying patios for anywhere between 2 to 6 weeks.
Chemical Changes During Roasting
The transformation from raw bean to finished product is perhaps one of the more complex stages in coffee production.
If you take a lot of green beans and smell it, it hardly has any of the characteristics that we would typically associate with roasted coffee. Yet once roasted, the raw materials within the raw bean undergo a significant transformation to give rise to hundreds of new compounds that we can appreciate.
Drum vs Fluid Bed Roasters
Unlike products other food products which require minimal amounts of thermal processing, coffee undergoes a dramatic chemical changes before it even exhibits any hint of its aromatic complexity. For this to occur, we must pass the coffee over the fire, so to speak, and transform this relatively boring bean into a bean with a myriad of complexity.





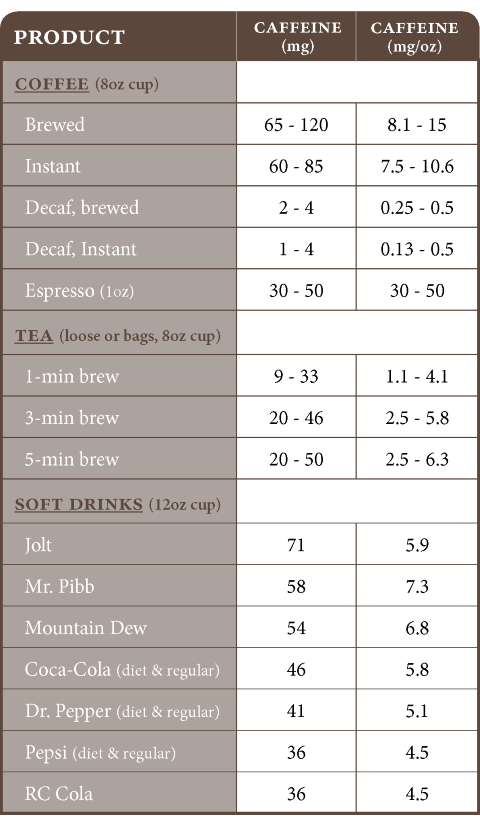 Source: NCA, August 1999, www.coffeescience.org
Source: NCA, August 1999, www.coffeescience.org