Displaying items by tag: bitterness
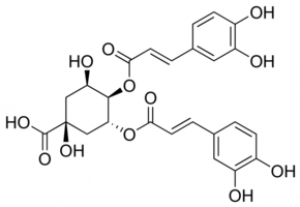
Chlorogenic Acid
Discovered in 1932, chlorogenic acids (CGA) represent a large family of esterified compounds present in green and roasted coffee. During roasting, CGA's slowly decompose to form caffeic and quinic acid with about 50% of the original CGA being destroyed in a medium roast.
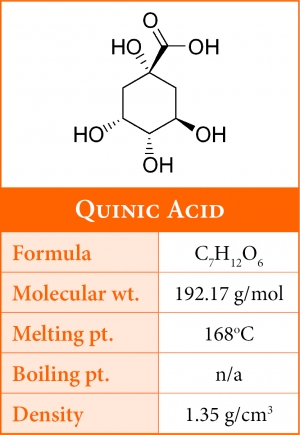
Quinic Acid
Quinic acid along with citric and malic represent a significant portion of coffee's total acid content. During roasting quinic acid progressively increases as the levels of chlorogenic acid decrease, suggesting that its formation results from the cleavage of the chlorogenic acid moiety.
Brewing Fundamentals
Unlike wine where the winemaker has complete control of the product up to the time of bottling, coffee requires that the consumers use a bit of their own skills during beverage preparation. This final step in coffee preparation is perhaps one of the most critical, since errors here can ruin the quality of the coffee and ultimately all the work that was invested into it.
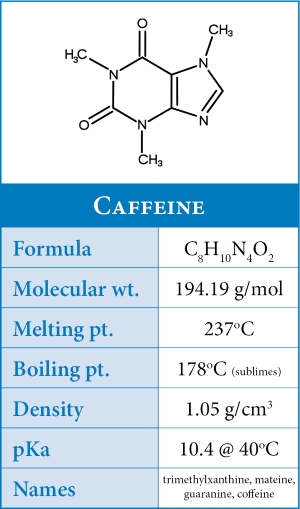
Caffeine in Coffee
In its pure state - caffeine, or 1,3,7-trimethylxanthine, exists as a white powdered alkaloid with an intensely bitter taste. Although coffee is most associated with caffeine, there are over 1,000 compounds in coffee making caffeine just a tiny fraction (~0.01%) of coffee's total composition.